
Molecules | Free Full-Text | Concise Syntheses of Marine (Bis)indole Alkaloids Meridianin C, D, F, and G and Scalaridine A via One-Pot Masuda Borylation-Suzuki Coupling Sequence
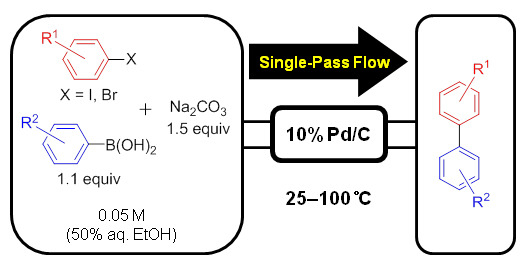
Catalysts | Free Full-Text | Palladium on Carbon-Catalyzed Suzuki-Miyaura Coupling Reaction Using an Efficient and Continuous Flow System

Palladium catalyzed asymmetric Suzuki–Miyaura coupling reactions to axially chiral biaryl compounds: Chiral ligands and recent advances - ScienceDirect

Palladium Extraction Following Metal-Catalyzed Reactions: Recent Advances and Applications in the Pharmaceutical Industry | Organic Process Research & Development

Palladium-Catalyzed Suzuki−Miyaura Cross-Coupling Reactions Employing Dialkylbiaryl Phosphine Ligands | Accounts of Chemical Research

Palladium-Catalyzed Suzuki−Miyaura Cross-Coupling Reactions of Potassium Aryl- and Heteroaryltrifluoroborates | The Journal of Organic Chemistry

Palladium-Catalyzed Suzuki−Miyaura Cross-Coupling Reactions Employing Dialkylbiaryl Phosphine Ligands | Accounts of Chemical Research

Palladium-Catalyzed Suzuki−Miyaura Cross-Coupling Reactions Employing Dialkylbiaryl Phosphine Ligands | Accounts of Chemical Research

Palladium-Catalyzed Suzuki−Miyaura Cross-Coupling Reactions Employing Dialkylbiaryl Phosphine Ligands | Accounts of Chemical Research

Palladium-Catalyzed Suzuki−Miyaura Cross-Coupling Reactions Employing Dialkylbiaryl Phosphine Ligands | Accounts of Chemical Research

Advances in Transition‐Metal Catalyzed Carbonylative Suzuki‐Miyaura Coupling Reaction: An Update - Bhattacherjee - 2021 - Advanced Synthesis & Catalysis - Wiley Online Library

Palladium-Catalyzed Suzuki–Miyaura Coupling of Aryl Esters | Journal of the American Chemical Society

Palladium Nanoparticles on Covalent Organic Framework Supports as Catalysts for Suzuki–Miyaura Cross-Coupling Reactions | ACS Applied Nano Materials

Palladium-Catalyzed Suzuki–Miyaura Coupling of Aryl Esters | Journal of the American Chemical Society
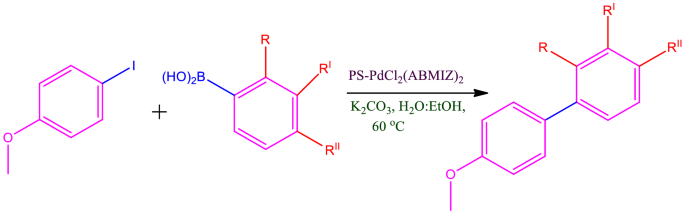
Suzuki–Miyaura Cross Coupling Reaction Using Reusable Polymer Anchored Palladium Catalyst | Catalysis Letters
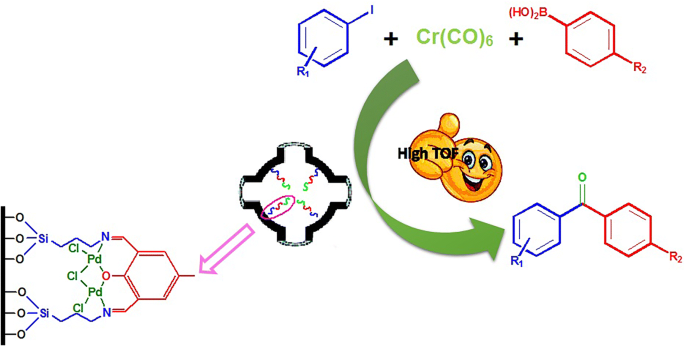
Binuclear Palladium Complex Immobilized on Mesoporous SBA-16: Efficient Heterogeneous Catalyst for the Carbonylative Suzuki Coupling Reaction of Aryl Iodides and Arylboronic Acids Using Cr(CO)6 as Carbonyl Source | Catalysis Letters

Recent Advances in the Palladium Catalyzed Suzuki–Miyaura Cross-Coupling Reaction in Water | Catalysis Letters

Homogeneous and Recyclable Palladium Catalysts: Application in Suzuki– Miyaura Cross-Coupling Reactions | Organometallics

Palladium-Catalyzed Suzuki−Miyaura Cross-Coupling Reactions Employing Dialkylbiaryl Phosphine Ligands | Accounts of Chemical Research
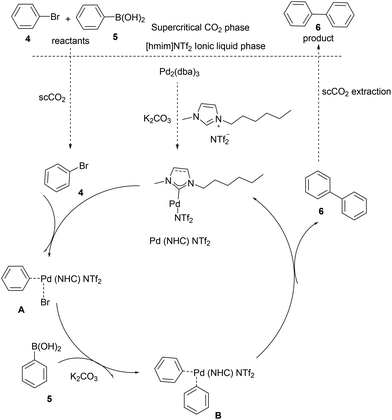
Recent advances in the Suzuki–Miyaura cross-coupling reaction using efficient catalysts in eco-friendly media - Green Chemistry (RSC Publishing) DOI:10.1039/C8GC02860E

Palladium-Catalyzed Suzuki−Miyaura Cross-Coupling Reactions Employing Dialkylbiaryl Phosphine Ligands | Accounts of Chemical Research

Molecules | Free Full-Text | Concise Syntheses of Marine (Bis)indole Alkaloids Meridianin C, D, F, and G and Scalaridine A via One-Pot Masuda Borylation-Suzuki Coupling Sequence

